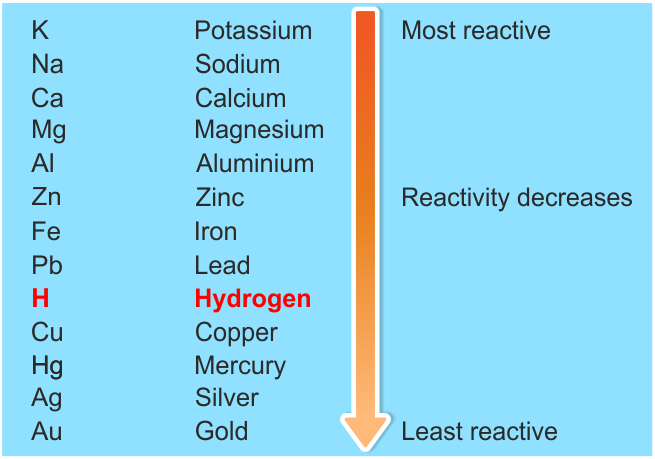
A few drops of universal indicator have been added to the water. Questionĭescribe and explain the observations when a small piece of lithium is placed on the surface of a big container of water. 2.1.1 recall the reactivity series of metals, including K, Na, Ca, Mg, Al, Zn, Fe and Cu 2.1.3 explain how the reactivity of metals is related to the tendency of a metal to form its positive ion 2.1. In general, the more reactive the metal, the more rapid the reaction is. Sodium + water → sodium hydroxide + hydrogen For example, sodium reacts rapidly with cold water: When a metal reacts with water, a metal hydroxide and hydrogen are formed. Hydrogen and carbon are shown for comparison. The table summarises some reactions of metals in the reactivity series. For example, both magnesium and zinc can react with hydrogen ions to displace H 2 from a solution by the reactions: Mg (s) + 2 H + (aq) H 2 (g) + Mg 2+ (aq) Zn (s) + 2 H + (aq) H 2 (g. The top metals are more reactive than the metals on the bottom. the more easily it loses electrons in reactions to form positive ions (cations) The activity series is a chart of metals listed in order of declining relative reactivity. 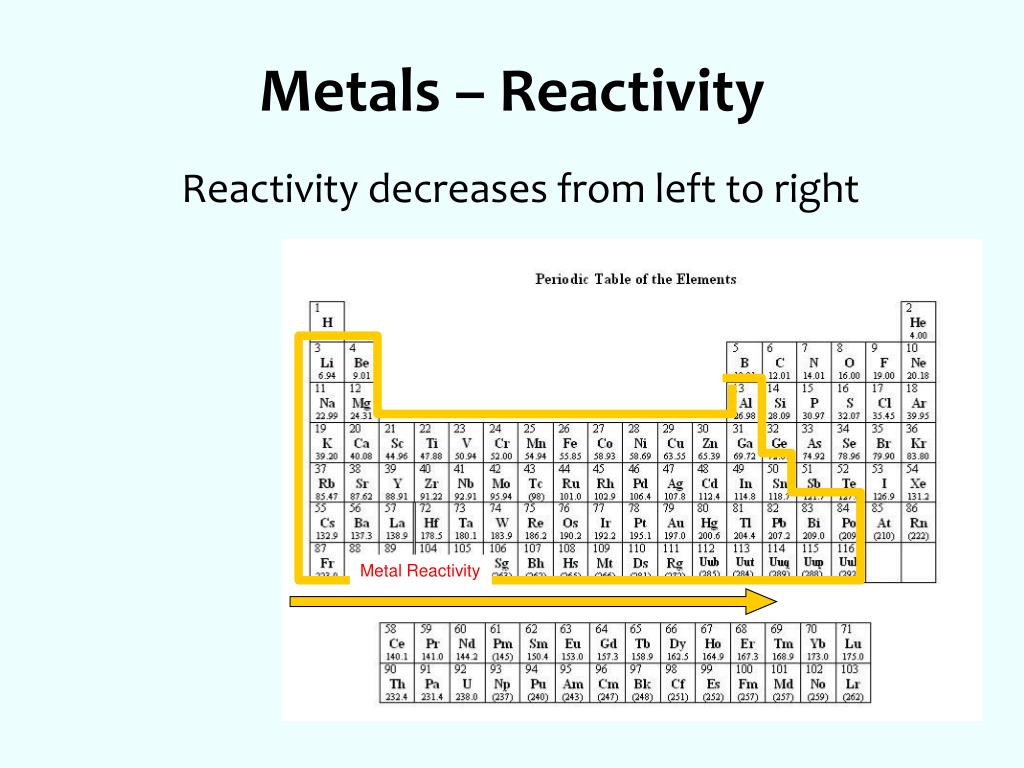
In general, the more reactive a metal is: The reactivity series of metals is a chart showing metals in order of decreasing reactivity. Metal displacement occurs when a more reactive metal forms a cation and displaces a less reactive metal from its compounds.ĥ.When metals react with other substances, the metal atoms lose electrons to form positive ions. This provides the driving force for metal displacement reaction. In other words, a highly reactive metal prefers to be a cation while its less reactive friend prefers to be a free element. A less reactive metal is a hoarder - it would rather have its valence electrons. The reactivity series tells us how much a metal wants to be a cationĪ more reactive metal donates electrons more readily to form a cation. More reactive metals react more vigorously, increasing the rate of effervescence of hydrogen gas.Ĥ. Secondly, reactivity affects the rate of reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
